|
Tendyne Succeeds in MAC, Pfizer vs. Novo, and Atorvastatin Recall
November 3, 2025
|
|
|

|
|
Together with
|

|
|
|
“New article in Lancet says that money has become the mission of U.S. healthcare, highlighting that healthcare workers are ‘demoralized by pressure to prioritize their employer’s financial goals over patient needs… as space for good work in a bad system narrows.’”
|
|
Laura Vater, MD, MPH
|
|
|
|
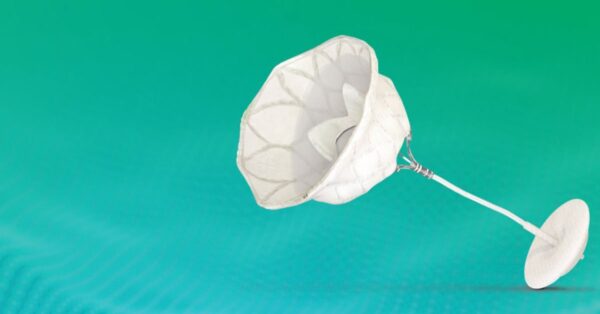
New research presented at TCT 2025 suggests that Abbott’s FDA-approved Tendyne TMVR system leads to significantly better health outcomes in patients with severe mitral annular calcification who were previously considered untreatable.
- Severe mitral annular calcification (MAC) with mitral regurgitation or stenosis is typically considered extreme and is excluded from interventional treatment due to technical challenges.
- The Tendyne system received FDA approval earlier in 2025 based on SUMMIT trial data, but its efficacy in severe MAC patients was unknown till now.
The SUMMIT-MAC trial enrolled 103 patients with severe MAC and found that the challenging patient population showed strong outcomes despite extreme pathology…
- The study’s primary composite endpoint of freedom from all-cause mortality and HF hospitalization at 12 months was 60.4%, significantly exceeding the 43% goal.
- Technical success reached 94.2% despite an average MAC volume of 5,679 mm³ (which is considered very extreme).
- Mitral regurgitation resolution was excellent, with 100% of patients having none/trivial MR at discharge, and 91.4% remaining regurgitation free at 12 months.
- Quality-of-life improvements were substantial, with KCCQ scores improving by 18.7 points on average.
However, patients treated with the Tendyne system still faced significant procedural challenges caused by MAC including some outcomes like:
- A 30-day mortality rate of 6.8% (all cardiac deaths) and a 12-month mortality rate of 21% – partially due to 85.4% of patients being significantly frail.
- Bleeding complications occurred in 27.2% of patients including 6.8% life-threatening bleeds – mostly due to Tendyne’s transapical access route.
- The study also had a 74.6% screen failure rate – so careful patient selection is critical as well as the complexity of MR patients with MAC.
Although U.S. guidelines currently do not offer any recommendations for TMVR in MAC patients, there’s a good chance the SUMMIT-MAC data will change this, with European societies already providing class 2b recommendation for TMVR in severe MAC.
The Takeaway
Thanks to SUMMIT-MAC, we now know that Abbott’s Tendyne TMVR system successfully treats severe mitral annular calcification, which is a big win for a patient population that is usually excluded from all interventional options.
|
|
Cracking the Code of ECG Analysis with Monebo’s AI Genetic Algorithm
How do you teach a computer to recognize the complex patterns in an ECG signal? Discover how Monebo found the answer by blending human expertise and a little evolutionary magic in this article that breaks down the advantages of genetic algorithms in ECG analysis.
|
|
Tempus Receives FDA 510(k) Clearance for Tempus ECG-Low EF
Tempus announces the expansion of its Tempus ECG-AI portfolio with Tempus ECG-Low EF, software intended for use to analyze 12-lead ECG recordings and detect signs associated with having a low left ventricular ejection fraction (LVEF less than or equal to 40%) in patients 40 years of age or older at risk of heart failure. It is not intended as a stand-alone diagnostic and positive results may suggest the need for further clinical evaluation. For Full Indications for Use, visit here.
|
|
The Hidden Costs of Delaying Cardiac MRI Adoption
Despite being the gold standard for functional cardiac imaging, many hospitals remain slow to adopt it. From missed revenue to lost patients, the impact goes far beyond the scan. Read about the real clinical and financial risks of falling behind.
|
|
- Philips iFR Helps With MI Decisions: Philips announced results from the iMODERN study at TCT 2025 that suggest there might not be a benefit to waiting to perform revascularization on acute MI patients. The study examined whether doctors should treat multiple blocked arteries during one procedure or wait for a second appointment and found that both approaches are equally safe and effective over three years. The findings also supported using Philips’ iFR technology to measure artery blockages during emergency MI procedures, helping decide the best treatment timing for individual patients.
- Novo Nordisk Outbids Pfizer for Obesity Drug: Novo Nordisk launched an $8.5B bid for obesity biotech developer Metsera, outbidding Pfizer’s initial $7.3B offer. Metsera called Novo’s proposal “superior,” giving Pfizer four days to negotiate. Pfizer then denounced the bid as “reckless” and anticompetitive, threatening legal action. Both companies are interested since Metsera is developing a once-monthly obesity drug that could differentiate it from current GLP-1 therapies. The battle reflects intense competition in the weight-loss drug market, projected to reach $150B by the early 2030s.
- Atorvastatin Recall: The FDA announced a recall of over 140k bottles of atorvastatin calcium (generic Lipitor), a cholesterol-lowering statin. The recall, classified as Class II, affects medication from Alkem Laboratories distributed by Ascend Laboratories. According to the FDA, the drug failed dissolution tests, meaning it doesn’t dissolve properly and may not effectively lower cholesterol. The recall was initiated in September and classified in October.
- Intellia Pauses Transthyretin Amyloidosis Trials: Intellia Therapeutics paused two clinical trials testing its Nex-z experimental gene-editing therapy for transthyretin amyloidosis, after a patient developed severe liver injury. The MAGNITUDE and MAGNITUDE-2 trials had enrolled over 650 and nearly 50 patients respectively, with approximately 450 receiving the treatment. Despite the pause, Intellia is already working with regulators to resume enrollment safely.
- Medline Makes it to Nasdaq: Surgical supplier Medline has officially filed paperwork with the SEC to go public after months of speculation. The company currently provides surgical devices and medical supplies globally, including cardiac and peripheral fluid management systems, vascular access systems, and various other OR and cath lab tools. The IPO could raise over $5B considering that Medline reported $13.5B in net sales with 9.7% revenue growth earlier this year. The company will trade on Nasdaq under the ticker MDLN.
- ATTR-CM and AEMD: A new study found that people with transthyretin amyloid cardiomyopathy (ATTR-CM) who have atrial electromechanical dissociation (abnormal upper heart chamber contractions) face over three times the stroke risk. Researchers analyzed over 2.3k patients and developed a risk-assessment tool using echocardiogram measurements to identify high-risk individuals, finding that 5% had AEMD, 33% had normal heart rhythm with effective left atrial contraction and, 62% had atrial fibrillation on anticoagulation.
- Smartwatch Algorithm for CVD Screening: Yale researchers developed an AI tool that can detect serious heart problems using a smartwatch’s built-in heart sensor. The algorithm, tested on 600 people, accurately identified structural heart diseases like weak pumping, damaged valves, or thickened heart muscle with 88% accuracy. Currently, these conditions require specialized ultrasound tests, so this technology could make heart disease screening more accessible since millions already wear smartwatches.
- Elucid’s Plaque IQ Expands to Carotid Analysis: Elucid expanded its FDA-cleared PlaqueIQ technology to plaque analysis in the carotid arteries. The AI technology identifies dangerous plaque types, including lipid-rich necrotic core, helping doctors assess stroke risk and personalize treatment plans. Unlike traditional methods that only measure artery narrowing, the PlaqueIQ software can evaluate both coronary and carotid arteries in a single scan, providing a comprehensive view of atherosclerotic disease to help prevent heart attacks and strokes.
- Orchestra and Terumo Break Up: Orchestra BioMed terminated its distribution agreement with Terumo for the Virtue Sirolimus AngioInfusion Balloon, used to treat coronary artery disease. As a result, Terumo will pay $10M for a right of first refusal and invest $20M in Orchestra stock. The agreement allows Orchestra to seek partnerships while giving Terumo review rights for 90 days after trial results. The Virtue balloon recently entered FDA trials comparing it to Boston Scientific’s Agent balloon, which is still the only approved drug-coated balloon for coronary use in the U.S.
- YorLabs’ Ultrasound Imaging System: YorLabs received FDA 510(k) clearance for its Intracardiac Imaging System ultrasound platform for heart procedures. The wireless system provides high-resolution real-time imaging for electrophysiology and interventional cardiology without requiring large upfront investments or dedicated equipment space. It features single-operator control to streamline workflows during procedures like ablation and left atrial appendage closure. The Oregon-based company plans a limited U.S. launch in 2026, targeting the rapidly growing intracardiac echocardiography market.
- A New Interventional Cardiology Company: Quebec-based Antegrade Medical closed a $5.2M seed financing round to support the development of minimally invasive tools for complex interventional cardiology procedures, including structural heart disease and cardiogenic shock treatment. Antegrade also plans to accelerate R&D and complete first-in-human studies with its antegrade approach technology. The new company will be led by CEO Louis Laflamme, who previously guided OpSens through its $345M acquisition.
|
|
5,600 Ways to Improve Your Cardiac Ultrasound Workflow
AI-powered measurements can enhance the way you acquire and interpret cardiac ultrasound. Learn how AI-powered ultrasound can help you overcome everyday limitations in echo. Read Siemens Healthineers’ white paper on how its AI software provides 5,600+ automated measurements to help improve workflow efficiency, consistency, and clinical confidence.
|
|
What Every Cardiologist Needs to Watch for in Heart Failure
Patients with preserved (HFpEF) or mildly reduced ejection fraction (HFmrEF) face a high risk of hospitalization for heart failure and cardiovascular death, even with guideline-directed medical treatment. Hear from Bayer’s Dr. Alanna Morris-Simon as she shares common heart failure signs to look out for.
|
|
From CPACS to CVIS
Cardiovascular imaging has come a long way from CPACS. Explore the evolution of cardiology image and data management in this Merge by Merative executive brief, and see what makes CVIS such a significant advancement.
|
|
- Explore Vitrea Advanced Visualization: Discover Canon Medical Healthcare IT’s suite of advanced imaging workflows designed to increase efficiency in cardiovascular imaging, and facilitate the assessment, diagnosis, and treatment of cardiovascular diseases. These cutting-edge tools support the delivery of faster, more accurate care while integrating seamlessly into clinical workflow.
- Relieving The Burden of Post-Processing: With the advent of advanced imaging technologies like CCTA come added burdens to technologists and diagnostic imaging centers. See how PIA can relieve the burden of post-processing, saving you time while helping your bottom line.
- The Largest Registry on Plaque Analysis in CAD: What if 50% of your CCTA patients could benefit from an adjustment to their treatment plan? Read more about Heartflow’s DECIDE registry that demonstrates how Heartflow Plaque Analysis using its Plaque Staging software empowers physicians with clinical insights that lead to real-world impact.
- Us2.ai’s AI HF Now Possible with Handheld Echo: The latest research shows Us2.ai’s software can take handheld echocardiography beyond its standard applications. Read this EHJ study about how swiftly and accurately Us2.ai’s HF detection software detects LVEF, closely matching expert human analysis of standard cart based echocardiograms.
- Circle CVI’s Cardiac CT Expansion: Watch this Cardiac Wire interview with Circle Cardiovascular Imaging’s Chief Product Officer, Scott Galbari as he sheds light on Circle CVI’s history with cardiac MRI and how the company is tackling the coming demand for cardiac CT.
|
|
|
|
|